PATIENT INFORMATION: Ear Implants
What … for? (Indications)
Ear implants help, when there is no understanding with speech using hearing aids. They are placed after middle ear surgery or malformation. Ear implants help at single-sided deafness.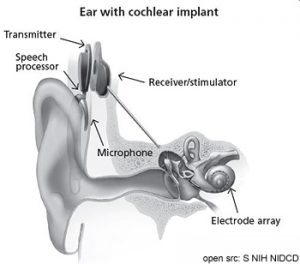
Numbers
Global: 219.000, USA 70.000 patients with cochlear implants. (ASHA). 1-3 newborns per 1.000 have a hearing loss that can be treated only with cochlear implants.
Times
Swelling around the wound for about 2-3 days.
Contraindications
behavioral
People who have been deaf for a short period of time do better with cochlear implants than people who have been deaf for long periods of time
medical
Minimum age is 18 years. There are special implants for 12-24 month old children. Middle ear infection. Perforation of the membrane tympanique due to an infection. Aplasia of the cochlea.
Risks
during surgery
- Nerv injury may lead to change of taste, rarely permanent.
- Injury to the facial nerv (temporary or permament weakening or full paralysis on the same side of the face occurs in <1 in 1000 cases.
- Leakage of brain fluid (~1%).
- 2-3% of patients will require their implant removed due to device malfunction or infection.
- General anesthesia necessary – relating risks.
short term
- Skin wound infection, cerebrospinal fluid leakage, numbness around the operation area,
long term
- Exposed implant, Meningitis (biofilm after microorganisms colonize boundary surfaces), perilymph fluid leak, chronic localized inflammation, reparative granuloma, tinnitus, vertigo or dizziness. Few reports on allergic reactions to silicone.
restrictions
- MRI scan not possible. Risk with ionic radiation therapy, electroconvulsive therapy, electrical surgery, and neuro-stimulation.
- Possible interaction with theft detection systems, metal detectors or other security systems.
- Possible affection by cell phones or radio transmitters.
- Possible Interaction in unpredictable ways with computer systems.
- CI-patients have to be careful of static electricity, something that can damage their implant.
- Skin irritation (rubbing of the external part).
- Strange sounds may be heard.
- Different hearing outcomes: A person might not hear as well as other people who experience successful outcomes with their implants.
- Potential loss of residual hearing: Cochlear implants might destroy any remaining hearing in the ear that has the implant.
Failures
Implant failures may happen and the person would have to undergo additional surgery
Material
External part
Microphone, sound processor, and transmitter system. They receive the sound, process it and send it to the internal part. This advice is necessary for the different types of ear implants.
Internal part
Implanted receiver and electrode system; something that contains electronic circuits that receive signals from the external system, and send electrical currents to the person’s inner ear. They activate the person’s nerve, sending a signal to their brain. The person’s brain learns to recognize the signal and the person experiences the signal as, ‘hearing.’
Inner parts differentiating
- Transmitter: Coil in a silicone sheath
- Electronics: Enclosed in a titanium casing
- Electrode: Platinum contacts in a silicone array
- Cable: Silicone, teflon
- Cochleostomy site
Electroacustic Stimulation
A cochlear implant combined with a hearing aid. For people to restore their hearing in the high frequencies, and acoustic amplification for the low frequencies
Middle Ear Implant
Vibrant soundbridge
Mechanical vibrations transmit the signals to the middle ear.
No cables to the inner ear necessary
Bone Conduction Implant
Bonebridge Implant
Mechanical vibrations in the temporal bone transmit the signals to the inner ear.
No cables to the inner ear necessary.
Statistics
USA: 36 million have hearing loss (one in every ten). Sudden sensoneural hearing loss 5-20 people per 100.000 persons
16% of Europeans require treatment. 50% over 65 have hearing loss. (MHH)
Literature
– Basic
– Problems
Wilson-Clark S: Bacterial Meningitis among cochlear implant recipients – Canada 2002
– Science/Future
Stöver T, Lenz T: Biomaterials in cochlear implants (also for basics on material)
Medical societies: see here
Criticism: n/a
Selected Patient Information
- Cochlear Implants: Facts, Benefits & Risks (2012)
- The Ear Foundation
- Cochlear Implants – Facts
- Cochlear Implants FDA
- Cochlea-Implantat oder Hörgerät (german)
- Cochlear implant candidacy criteria
- How the bone bridge, the active bone conduction implant works
Register for Patients
In the register of the registries you can check which ones are available.
The Implant-Register offers registration of implants online and you can download a printable version for your personal use.
Disclaimer: The information and links and whatsoever shown on this page are compiled with care. However, Implant-Register can´t take any responsibility for the information given, nor their content, nor their up-to-date nature, particularly in interlinked pages. You may help us with your contribution, granting us the decision to publish or not. Be careful with conclusions for yourself, in doubt double-check and consider medical solutions are individual and have to be found with an educated medical person.