AMS Ambicor™ Penile Implant
IMPLANT PROFILE |
||||||||
 |
unregistered register here |
|||||||
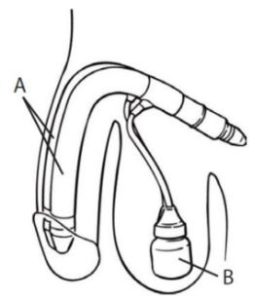 |
A. Cylinder B. Pump buld |
||||||
| the implant |
| 01 | indication | implant, to make the penis stiff and rigid | ||||||||
| 02 | specific | intended for use in the treatment of chronic, organic, male erectile dysfunction (impotence) | ||||||||
| 03 | contraindication(s) |
|
||||||||
| 04 | implant description | the implant is a fluid-filled device that includes a pair of cylinders implanted in the penis and a single pump located in the scrotum. squeezing and releasing the pump moves fluid to the cylinders, creating an erection. |
||||||||
| 05 | specific attributes | ◊◊◊◊◊ | ||||||||
| 06 | material(s) | solid silicone elastomers, fluorosilicone lubricant | ||||||||
| 07 | implant surface | n/a | ||||||||
| 08 | key literature | ◊◊◊◊◊ | ||||||||
| 09 | notes about the company | n/a | ||||||||
| 10 | approvals | CE-Marking | FDA-Approval | ||||||
| learn more about approvals, certifications, authorities | ||||||||
| No comments yet. You are welcome to send comments or experiences. HERE, if you received this implant. and HERE, if you placed this implant. |
||||||||
| Received this implant? You can register it HERE (fee). Placed this implant? You can register it HERE (fee). |
||||||||
| current as of 2022-10-25 | ||||||||
| unregistered / this page is not approved by the provider / unregistered implants have no professionnal lines or advertising | ||||||||
| This “Implant Profile” shows a non-registered implant. This is a most comprehensive description of the important parameters of an implant. Information given here are checked thoroughly by “Implant-Register” and is not approved by the manufacturer/the company. Illustrations refer to the company, if not shown differently. However we can not guarantee for any material or properties. Implants may not be available in all countries. If there are pictures like logos or implants, they are taken from the companies website. The publishing company may inform us to hide them. | ||||||||
